Learn more about the research of the Structure and Properties of Complex Materials Department.
Atomic structure and inhomogeneities in complex semiconductor materials
In this group, we study the correlation between structure and properties in complex semiconductors on a micrometer to subnanometer scale. The modern, multinary compounds under investigation are used, for example, in thin film solar cells and numerous electronic and optoelectronic devices. A main focus of our work is the determination of the element-specific atomic-scale structure and its influence on important material properties.
Furthermore, structural and compositional inhomogeneity in the nano- to micrometer range often plays a crucial role for the device performance. XRF KesteriteA comprehensive understanding of the correlation between preparation conditions, chemical composition and structure as well as electrical and optical properties is indispensable in order to utilize the potential of these material systems even more efficiently in the future. To that end, we apply different, typically synchrotron based X-ray techniques and various electron microscopy methods and we closely collaborate with a large number of national and international research groups.
Complex semiconductor alloys are applied in numerous electronic and optoelectronic devices ranging from light emitting diodes and laser over radiation detectors and transistors to high-efficiency thin film solar cells.
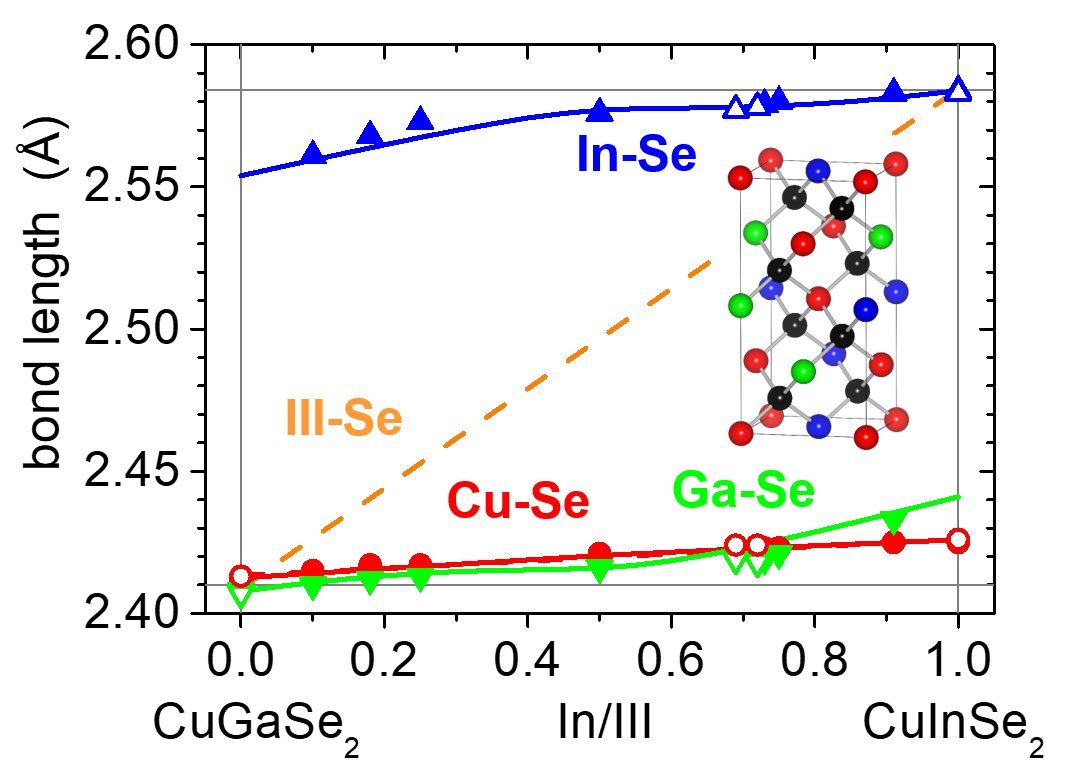
The properties of these materials can be specifically tailored by adjusting the alloy composition. Interestingly, the local atomic arrangements of lattice-mismatched semiconductor alloys typically exhibit a striking deviation from the long-range crystallographic structure.
This inhomogeneity on the subnanometer scale can also have a strong impact on the material properties. Therefore, the determination of element-specific local structural parameters, such as bond lengths and atomic displacements, is a major focus of our work.
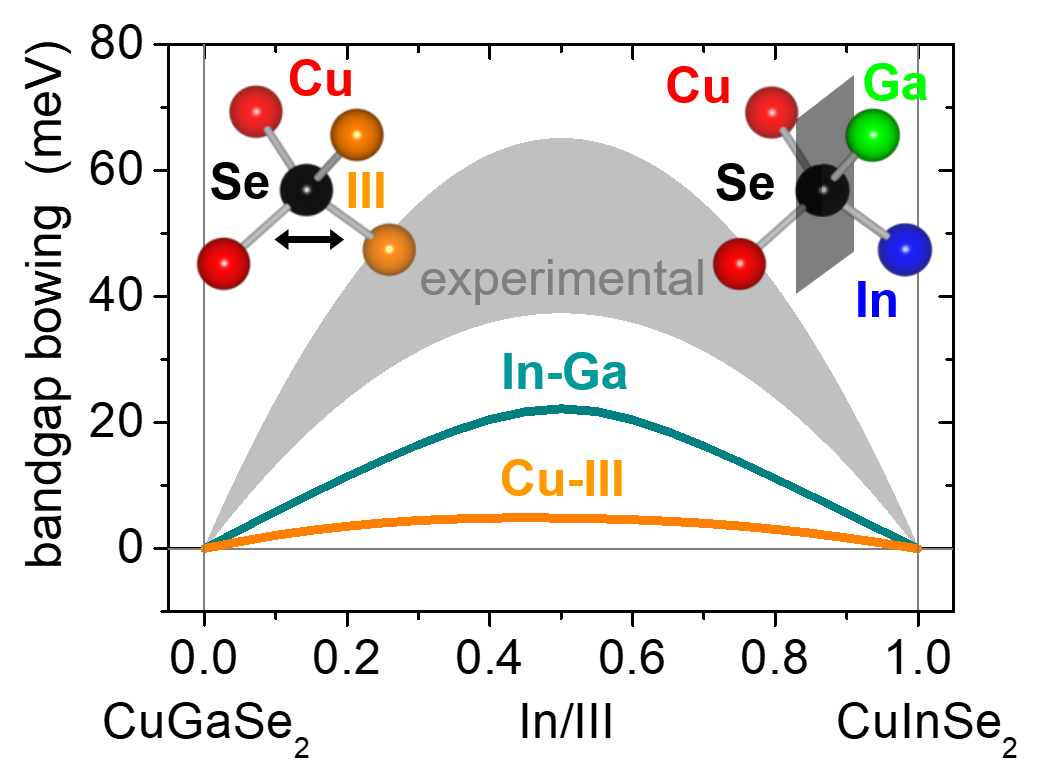
Band Gap BowingWe achieve this goal by using X-ray absorption spectroscopy. Furthermore, we investigate the correlation of these structural parameters with other material properties, most prominently the band gap energy or element-specific local electronic states. Among the materials studied in our group are chalcopyrites, like Cu(In,Ga)Se2 and Cu(In,Ga)S2, so-called kesterites, like Cu2ZnSn(Se,S)4, Cu2(Zn,Fe)SnS4 and Cu2Zn(Sn,Ge)Se4, and transition metal doped In2S3.
Compound semiconductor alloys: From atomic-scale structure to bandgap bowing
C. S. Schnohr
Applied Physics Reviews 2, 031304/1-43 (2015)
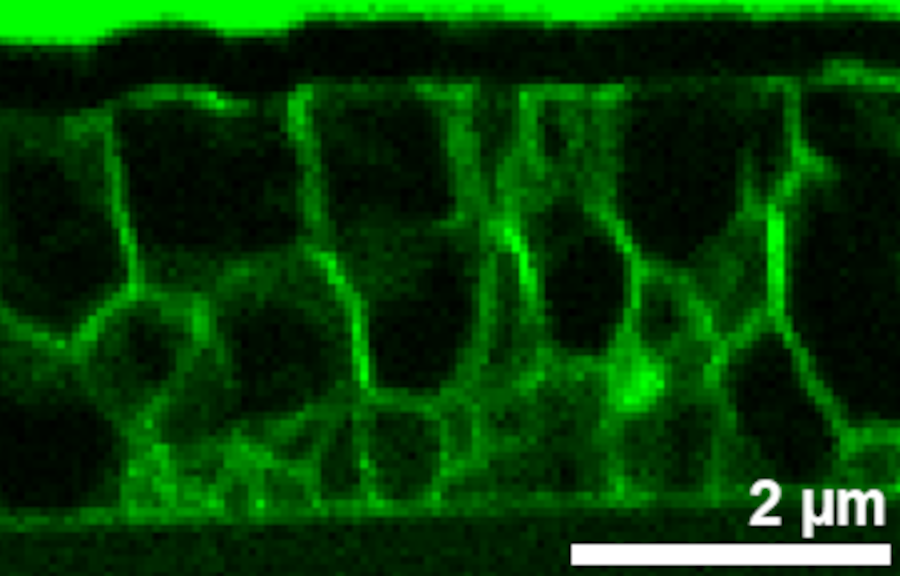
Polycrystalline thin film solar cells have achieved record efficiencies of more than 22%. These materials are characterized by a pronounced structural and chemical inhomogeneity on the nano- to micrometer scale, which can have either beneficial or detrimental effects on the solar cell performance.
Therefore, we study chemical inhomogeneity, in particular gradients and fluctuations of the composition, agglomeration of certain key elements and secondary phase segregation, using spatially resolved X-ray fluorescence analysis and correlate these with morphological features such as grain boundaries and interfaces, which we characterize by electron microscopy based techniques.
We investigate complete thin film solar cells based on chalcopyrites, like Cu(In,Ga)Se2, and so-called kesterites, like Cu2ZnSnSe4 and Cu2ZnSn(Se,S)4.
Rubidium segregation at random grain boundaries in Cu(In,Ga)Se2 absorbers
P. Schöppe, S. Schönherr, R. Wuerz, W. Wisniewski, G. Martínez-Criado, M. Ritzer, K. Ritter, C. Ronning, C. S. Schnohr
Nano Energy 42, 307-313 (2017)
Discrepancy between integral and local composition in off-stoichiometric Cu2ZnSnSe4 kesterites: A pitfall for classification
Ph. Schöppe, G. Gurieva, S. Giraldo, G. Martínez-Criado, C. Ronning, E. Saucedo, S. Schorr, C. S. Schnohr
Applied Physics Letters 110, 043901/1-5 (2017)
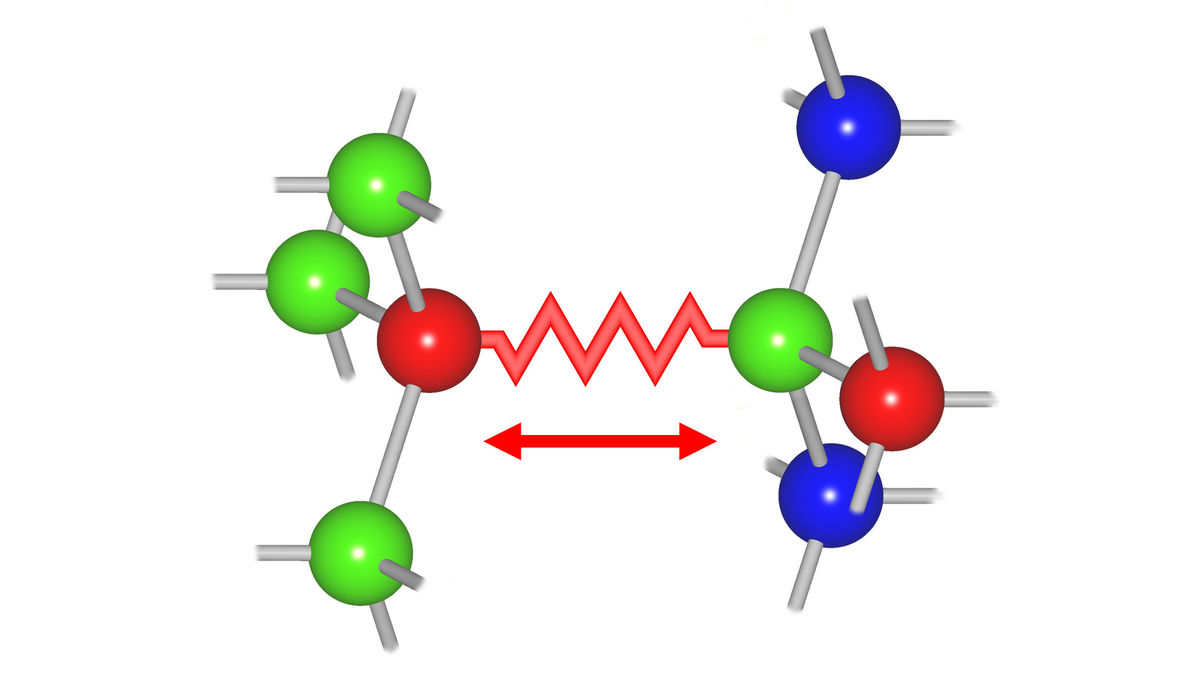
The thermal vibrations of atoms in a material are governed by the force constants of the interatomic bonds, which describe the resistance to stretching and bending of the bond.
These force constants also describe the response of a material to stress and strain and, hence, play a crucial role for the local atomic structure at interfaces between materials with different lattice constants, in nanoparticles and in semiconductor alloys.
We therefore apply temperature-dependent X-ray absorption spectroscopy to investigate the element-specific force constants of ternary semiconductor alloys as a function of the material composition.
The results provide unique insight into the complex vibrational behaviour of these important mixed systems and contribute to a better understanding of their electronic properties. We study ternary zincblende alloys, like (In,Ga)As and (In,Ga)P.
Bond-strength inversion in (In,Ga)As semiconductor alloys
S. Eckner, K. Ritter, P. Schöppe, E. Haubold, E. Eckner, J. Rensberg, R. Röder, M. C. Ridgway, C. S. Schnohr
Physical Review B 97, 195202/1-6 (2018)